Zephyrus Innovations said its new device— reportedly the first direct injection CSTD for subcutaneous and intramuscular administration—prevents splattering, spraying, and aerosolization of drugs.
VANCOUVER, B.C.—Zephyrus Innovations, a privately-owned medical device company that designs and manufactures safety syringes and closed system transfer devices (CSTDs), received product 510(k) Marketing Clearance from the U.S. Food and Drug Administration (FDA) for its VaporShield ™ Closed System Transfer Device, the company said in a December release.
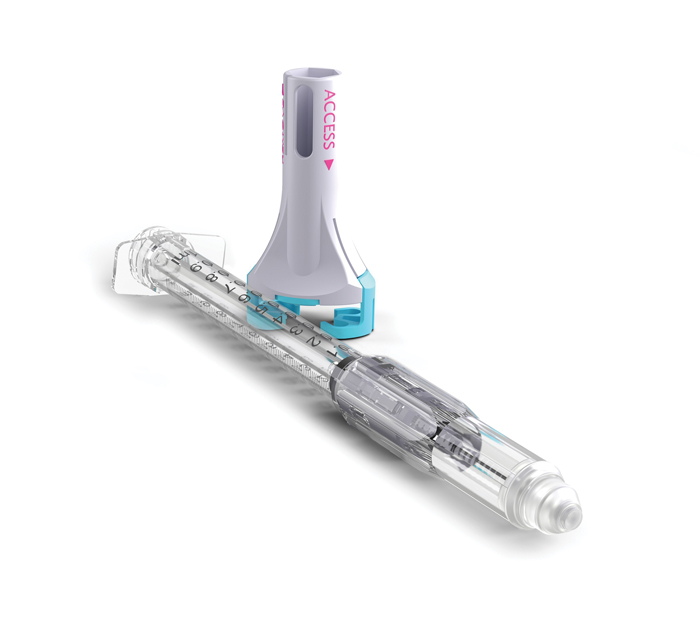
The VaporShield System comprises the VaporShield Closed System Transfer Device and drug vial holder (DVH). (Image courtesy Zephyrus Innovations)
The VaporShield™ Closed System Transfer Device is the latest innovation in a portfolio of safety syringes that Zephyrus Innovations is developing. According to the company, its safety syringes and closed system transfer devices deliver medication effectively while protecting clinicians from harmful drug exposure and needlestick injuries, which can lead to infection with bloodborne pathogens and serious disease.
Closed system transfer devices are critical to protecting healthcare workers from hazardous drug exposure, a protection that is now required in the United States under USP<800> legislation. No options currently exist that can meet this legislation for healthcare workers who deliver subcutaneous or intramuscular injections, the release stated.
According to Zephyrus, VaporShield is the first direct injection CSTD for subcutaneous and intramuscular administration. The company describes the device as “an end-to-end closed system” that prevents “splattering, spraying, and aerosolization of drugs.” VaporShield reportedly provides a true closed system from drug draw to injection and through to disposal, while retaining the crucial safety syringe features of Zephyrus’s Aeroject™ auto-retractable syringe to prevent needlestick injuries.
“VaporShield is a truly game-changing device,” said Guy Reynolds, executive chairman of Zephyrus Innovations, in a statement. “The seriously negative health impacts caused by exposure to hazardous drugs are well documented, but unfortunately, there has been no device that fully protects healthcare workers giving these drugs via subcutaneous or intramuscular administration, until now. There is now a significant opportunity to protect millions of healthcare workers around the world as they perform their day-to-day jobs.”
The global market for closed system transfer devices is expected to reach approximately $2.7 billion by 2030, the release stated, with the U.S. accounting for 40 percent of sales.
“The development of this device and its clearance has been a remarkable team effort, drawing on the extensive experience that we have across the business,” Reynolds stated. “I would like to thank them, along with our shareholders, for their commitment and support to date.”
