Medtronic’s next-generation platform is cleared for spine procedures in the U.S. and is designed to support future cranial and ENT applications, pending 510(k) clearance.
GALWAY, Ireland—Healthcare technology company Medtronic reported in February that it received U.S. Food and Drug Administration (FDA) clearance of the Stealth AXiS™ surgical system. The next-generation platform brings “planning, navigation, and robotics together into a single, intelligent system for spine surgery,” the company said in a release.
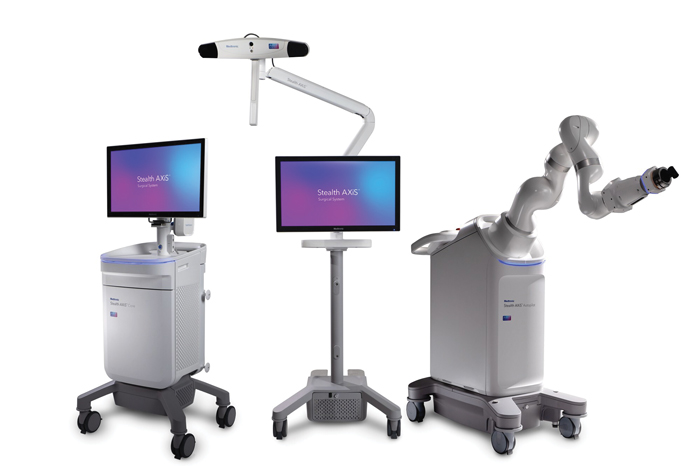
Medtronic’s Stealth AXiS™ surgical system. (Image: Medtronic/PRNewswire)
The Stealth AXiS system is cleared for spine procedures in the United States. Its underlying architecture is designed to support future cranial and ENT applications, pending 510(k) clearance.
“Built to support a wide range of surgeon preferences, clinical complexity, and care settings, the platform is designed for use across hospitals and ambulatory surgery centers without relying on multiple standalone technologies,” the release stated.
According to Medtronic, its Stealth AXiS™ system is grounded in more than 50 years of the company’s leadership in surgical navigation and robotics. It is said to create a clear pathway for adoption by combining familiar navigation workflows with a modular robotic design, allowing institutions to deploy what they need today and expand over time as clinical needs evolve.
“The Stealth AXiS™ system represents a meaningful step forward in how we think about navigation and robotics working together,” said Ronald A. Lehman, Jr., M.D., tenured professor of orthopaedic surgery (neurological surgery) at Columbia University, division chief of spine surgery and spine medical director at The Spine Hospital at NewYork-Presbyterian/The Allen Hospital, and a paid consultant to Medtronic, in the release.
“One of the biggest challenges in spine surgery has been understanding and responding to how the spine moves during a procedure. The Stealth AXiS™ system gives surgeons real-time visibility into that motion, helping us more consistently achieve our surgical plan without interrupting workflow. To me, this is truly game changing technology.”
A key innovation of the Stealth AXiS™ system is LiveAlign™ segmental tracking. It is reportedly an industry-first capability that allows surgeons to visualize anatomic motion, surgical adjustments, and patient alignment in real time during spine surgery, without the need for repeated imaging.
“This capability helps reduce reliance on manual steps and workflow disruption, supporting more consistent execution of patient-specific surgical plans,” the release stated.
As a cornerstone of Medtronic’s AiBLE™ smart ecosystem, the Stealth AXiS™ system is said to enable a more intuitive and seamless flow of information across the surgical continuum. By natively integrating planning, navigation, and execution within a single platform, it helps streamline workflows and supports the exchange of insights before, during, and after spine surgery. This allows the broader AiBLE™ ecosystem to connect devices, software, and data in a more meaningful way, the company said in the release.
“Spine surgery is complex, and variability remains a real challenge,” said Michael Carter, senior vice president and president of Medtronic Cranial & Spinal Technologies, in the release. “The Stealth AXiS™ system is designed to make advanced technology more usable and clinically meaningful, helping surgeons deliver more predictable, personalized care while laying the foundation for continued innovation.”
The Stealth AXiS™ surgical system is said to reflect Medtronic’s focus on building durable platforms that “simplify surgical workflows today while enabling future expansion across procedures, specialties, and care settings.”
