The U.S. FDA granted clearance to a new system, created by iotaMotion, that is said to preserve delicate intracochlear structures.
ST. PAUL, Minn.—A robotic-assisted cochlear implant insertion system developed by medical technology company iotaMotion, Inc., has received U.S. Food and Drug Administration (FDA) 510(k) clearance for expanded pediatric use.
In a January release, iotaMotion stated that the FDA cleared its iotaSOFT® Insertion System for use in patients four years of age and older, extending access to robotic-assisted cochlear implantation for school-aged children.
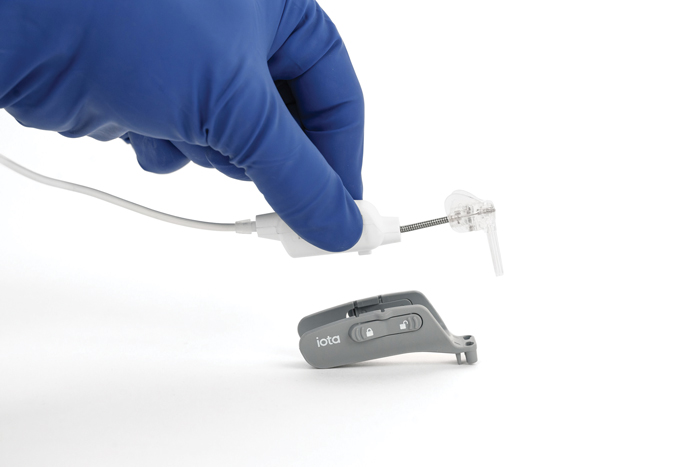
The iotaSOFT® Robotic-Assisted Cochlear Implant Insertion System is reported to advance surgery beyond the limits of the human hand with slow, consistent, and controlled electrode insertion to preserve delicate intracochlear structures. Shown with the drive unit guiding the array, iotaSOFT is FDA cleared for patients 4 years and older, expanding access as fewer than 5 percent of 60 million eligible patients worldwide receive implants. (Image: iotaMotion, Inc./PRNewswire)
“Receiving FDA clearance for expanded pediatric use marks a significant milestone for iotaMotion and for families navigating cochlear implant decisions,” said Mike Lobinsky, CEO of iotaMotion, in the release. “By standardizing one of the most delicate steps in cochlear implant surgery, iotaSOFT helps reduce variability, which could provide parents greater confidence as they consider cochlear implantation for their children.”
The iotaSOFT Insertion System is reported to enable precise and controlled electrode array insertion during one of the most delicate steps of cochlear implant surgery. By standardizing this critical step, iotaSOFT is designed to help preserve delicate cochlear structures, a consideration that is often central to decision-making.
As part of this milestone, Cincinnati Children’s Hospital, a nationally ranked pediatric medical center, and a U.S. News & World Report Honor Roll member for 2025–2026, has become the first dedicated pediatric center to adopt the iotaSOFT Insertion System. Cincinnati Children’s joins more than 35 leading cochlear implant centers across the United States, including nearly half of U.S. neurotology fellowship training programs, that have adopted the innovative technology.
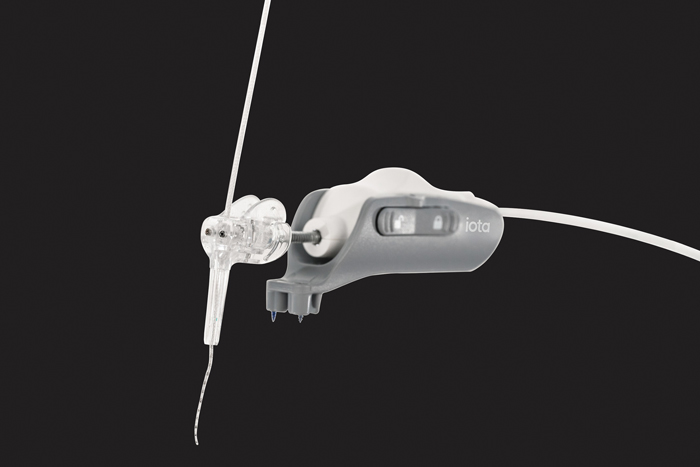
A close-up view of the iotaSOFT® Robotic-Assisted Cochlear Implant Insertion System. The system is FDA cleared for patients four years of age and older. (Image: iotaMotion, Inc./PRNewswire)
“At Cincinnati Children’s, our focus is delivering the highest standard of care while thoughtfully integrating innovations that benefit our patients,” said Dr. Daniel Choo, chief clinical growth officer and professor of otolaryngology–head and neck surgery. “Robotic-assisted cochlear implantation represents a meaningful advancement in our cochlear implant program.”
Although more than 430 million people worldwide experience disabling hearing loss, fewer than 5 percent of eligible cochlear implant candidates receive an implant, according to the World Health Organization. In pediatric patients, early access to sound is critical for speech, language, and educational development, yet concerns around hearing preservation and surgical variability can delay intervention, the release stated.
A 2025 clinical cohort study published in The Laryngoscope (Khan, et al.) reportedly found that 85 percent of patients in the robotic-assisted insertion group maintained hearing preservation at one year, compared with 71 percent in the manual insertion group, according to its authors.
“Robotic assistance in cochlear implantation is about precision and consistency,” said Marlan Hansen, M.D., co-founder and chief medical officer of iotaMotion, in the release. “With the expanded pediatric indication for iotaSOFT, supported by growing clinical evidence, families can have greater confidence that controlled, standardized insertion is designed to protect the cochlea and preserve its structure and function. This is especially important for children who may benefit from emerging therapeutic advances, including gene-based and regenerative hearing technologies, which will likely depend on atraumatic, cochlear implant array placement early in life.”
IotaMotion, Inc., based in St. Paul, Minnesota, is dedicated to advancing cochlear implant surgery beyond human capability through the development of robotic-assisted technology. The company’s flagship technology, the iotaSOFT® Insertion System, is designed to preserve delicate intracochlear structures through slow, consistent, and controlled electrode insertion. The system is commercially available in the United States and under clinical investigation in other global markets.
