Expanded Platform to Support Posterior-Based Surgical Approaches in Total Hip Arthroplasty
OrthAlign’s software enhancement enables surgeons to use the platform with their preferred surgical approach, including posterior-based approaches.
IRVINE, Calif.—Medical device company OrthAlign, Inc., a developer of surgical navigation technologies, recently expanded its Lantern® Hip platform to include support for posterior-based approaches in total hip arthroplasty (THA). By significantly broadening the platform’s clinical utility, the enhancement is said to enable surgeons “to choose Lantern Hip for any THA procedure, regardless of their clinical preference for patient position,” whether lateral or supine.
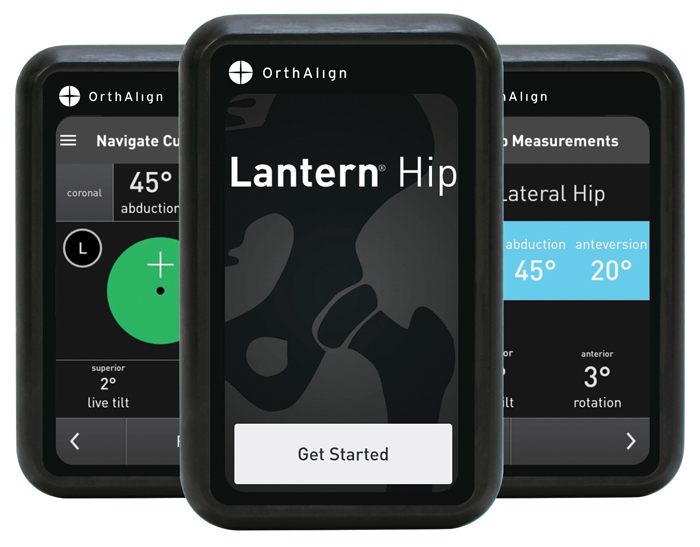
The new software increases access to Lantern Hip’s handheld, imageless navigation technology.
Lantern Hip’s portable design, paired with its clinical accuracy and precision, are said to make it an attractive alternative to large-footprint robots. (Photo courtesy OrthAlign, PRNewswire)
Because the majority of U.S. hip surgeons operate through posterior-based approaches, the new software increases access to Lantern’s handheld, imageless navigation technology.
“Lantern Hip is designed to deliver precise, real-time guidance that supports accurate implant positioning and reliable leg length restoration,” the release stated. “For patients in the lateral position, the surgeon can personalize cup position in the coronal plane according to each patient’s unique anatomy, with live pelvic tracking that accounts for any pelvic movement during the procedure. For patients in the supine position, the surgeon can compare cup position across the Anterior Pelvic Plane (APP) and Functional Pelvic Plane (FPP), as well as measure change in offset.”
The first clinical case to use the new “lateral” workflow was successfully completed on December 16, 2025, by John McLaughlin, DO, Orthopedic Surgeon at the Cleveland Clinic, according to OrthAlign.
“The addition of posterior-based approaches to Lantern Hip is an important step forward in supporting surgeon choice without compromising accuracy,” McLaughlin said in a statement. “In my first case, the system provided the same intuitive workflow and confidence in implant positioning that Lantern is known for. Having a compact, reliable navigation tool that adapts to my preferred surgical approach and implant enhances both efficiency in the OR and consistency for my patients.”
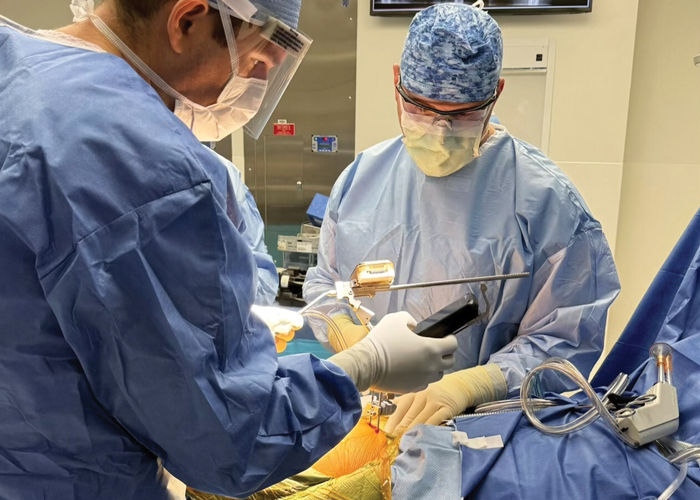
Orthopedic surgeon John McLaughlin, D.O., uses OrthAlign’s Lantern® Hip platform during an operation. (Photo courtesy OrthAlign, PRNewswire)
Lantern Hip is described as “an accessible option for any hospital or ASC.” Its capital-free model and portable design, paired with its clinical accuracy and precision, are said to make it an attractive alternative to large-footprint robots. Its instrumentation is compatible with most implant systems and surgical approaches, empowering surgeons to choose the best implant and surgical plan for each patient without vendor-imposed restrictions, the company said in the release.
“By accommodating posterior-based surgical approaches, OrthAlign now has the ability to support any surgeon performing total hip arthroplasty,” said OrthAlign CEO Eric Timko, in a statement. “This expansion reflects our commitment to delivering high-value, user-friendly technologies that improve accuracy and accessibility for surgeons everywhere.”
Lantern Hip initially launched for the supine patient position in 2023 and is expected to be fully commercially available for the lateral patient position in 2026.
OrthAlign describes itself as a medical device company that is focused on delivering practical, cutting edge technologies for orthopedic surgery. Committed to innovation and excellence, the company aims to provide surgeons with user-friendly, cost-effective solutions to improve patient care in joint replacement. According to OrthAlign, it continues to “advance surgeon-centric, handheld navigation technologies that reduce complexity and contain costs while enhancing accuracy in total joint arthroplasty.”
“Driven by the belief that everyone deserves exceptional healthcare, we are committed to making empowering technologies accessible to all,” the company stated in the release.
Lantern is a registered trademark of OrthAlign, Inc.
Titanium Knee Resurfacing System Is Designed to Treat Early Osteoarthritis, Cartilage Defects
Overture Orthopaedics developed a new system that was recognized by Octane with a 2025 High Tech Award for Best Innovation in Medical Devices.
IRVINE, Calif.—Overture Orthopaedics is a privately held, U.S.-based medical device company that provides surgeons with innovative joint preservation implants for use in sports medicine and orthopedic surgery. These specially designed implants are intended to relieve pain and dysfunction associated with articular cartilage lesions, osteochondral defects, and early osteoarthritis.
“We believe that our solutions raise the standard of care in joint preservation by providing state-of-the-art surgical tools that streamline use in the operating room, reduce the global cost of care, and enhance outcomes so that patients can get back to and maintain their active lifestyles longer,” Overture stated in a company release.
Last July, the company rolled out its OvertureTi Knee Resurfacing System with a full commercial launch in the United States. Three months later, the system was recognized as the best innovation in the medical device category by a Southern California organization that works to promote innovation in the medical device technology ecosystem.
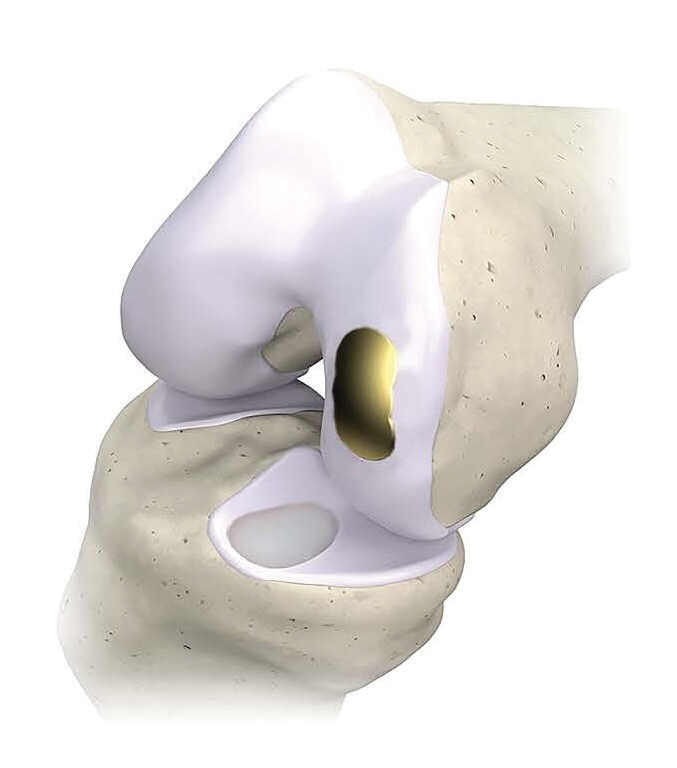
Femoral and tibial components of the OvertureTi Knee Resurfacing System®. The implants’ baseplates are made of 3D-printed, porous titanium to promote osseointegration and solid fixation. Articulating surfaces for the femoral and tibial components are titanium nitride and an overmolded vitamin E-treated, highly crosslinked polyethylene, respectively. (Image courtesy Overture Orthopaedics, PRNewswire)
The knee resurfacing system is composed of femoral and tibial implants that are intended to be used in partial replacement of articular surfaces of the knee, according to a release accompanying the launch. The implants were designed with sizing options that allow surgeons to replace only the diseased or damaged region of the joint—while preserving healthy surrounding cartilage and meniscus—in a procedure known as Focalplasty®.
“My career has been defined by osteochondral allografts,” stated Riley J. Williams III, M.D., chief of the Sports Medicine Institute at the Hospital for Special Surgery in New York City, in the release. “At some point, biology fails and no longer becomes a good option for a patient, but it’s far too early for any kind of joint replacement. Overture is bridging that gap between biologic options and joint replacement with an implant that resurfaces the joint, providing a durable, predictable, and lasting solution that adheres to the principles of joint preservation.”
With an ever-increasing population of active patients, the number of cartilage restoration surgeries performed annually is reported to exceed 300,000. A 2021 study on chondral lesions of the knee, conducted by Dekker et al., and published in the Journal of Bone and Joint Surgery, points out that current surgical and biological options for treating isolated cartilage defects continue to present patients and surgeons with significant challenges relating to costs and complexity. Among these challenges are inconsistent healing, donor site morbidity, and degradation over time, the release said.
Another study, by McCormick, et al., focused on trends in the surgical treatment of articular cartilage lesions in the United States. According to the study, some procedures, such as chondrocyte implantation, reach costs as high as $83,073 due to multi-stage surgeries, laboratory processing, associated episodes of care, and societal considerations.
According to Overture Orthopaedics, the OvertureTi Knee Resurfacing System implants are designed specifically as an alternative to be used when biological repair options are not viable for treating focal cartilage lesions.
The baseplates of the implants are 3D printed of porous titanium to promote osseointegration and solid fixation. Articulating surfaces for the femoral and tibial components incorporate titanium nitride and an overmolded, vitamin E-treated, highly crosslinked polyethylene, respectively. The implants are a fraction of the cost of biological options, the company said in the release.
“Overture is taking the concept of joint replacement—which has decades of research behind it, using a metal implant that gets lightly cemented and fixed into place, stable at time zero, and incorporates quite easily—then applying it to principles of joint preservation that we value in sports medicine,” said Kristofer J. Jones, M.D., associate professor of orthopedic surgery and sports medicine at UCLA and head team physician and director of orthopedic surgery for the Los Angeles Lakers, in the release.
“The concept is great because patients can get back to their active lifestyles quickly, they don’t have any downtime when it comes to weight bearing and range of motion restrictions, and they can readily start physical therapy to strengthen and recover, at time zero,” Jones continued. “We are also preserving the soft tissues around the knee.”
At the time of the full commercial launch in July, approximately 150 cases had been completed successfully in a limited market release that began in November 2023. The OvertureTi Knee Resurfacing System received 510(k) clearance from the United States Food and Drug Administration in March 2023, the release stated.
“We launched our limited market release to a select number of surgeons just over 18 months ago,” said James Young Kim, Overture’s CEO, in the July 2025 release. “The goal was to observe early patient outcomes, refine the surgical technique, and better understand patient selection. With a good number of cases under our belt and seeing how well our patients have been doing, we are ready for the full commercial release of the OvertureTi Knee Resurfacing System. We are excited to get the product in the hands of surgeons, so that they can better address knee pain and extend active lifestyles, ultimately raising the standard of care for patients.”
Overture Orthopaedics announced in October that it received the 2025 High Tech Award for “Best Innovation in Medical Device” from Octane, a Newport Beach, California-based organization dedicated to driving innovation in the medical technology and life sciences industries. The annual High Tech Award celebrates and recognizes a new standout medical technology that is expected to make a significant impact in the healthcare space and raise the standard of care for patients and surgeons.
“Joint preservation is what we’ve been looking for since day one,” said Jorge Chahla, M.D., associate professor of orthopedic surgery at Rush Medical Center in Chicago, and team physician for the Chicago Bulls and Chicago White Sox, in a statement. “Overture has a very minimally invasive approach; you don’t have to take out any unnecessary cartilage and bone, which is a great thing because you are preserving most of the cartilage and preserving most of the bone in the event the patient needs a replacement in the future.
“The other important thing is you are not touching any of the ligaments, which we know play a very important role in proprioception, making people feel more normal in their knee. You can achieve normal native biomechanics with Overture, which can be a game changer in joint preservation.”
“Being recognized by Octane and the greater community is a rewarding moment for us here at Overture,” said Overture Orthopaedics Chief Executive Officer James Young Kim, in a statement. “This recognition reflects the innovation behind our mission to raise the standard of care in joint preservation. We are confident that we are taking the small steps necessary to rethink treatment of the knee, where less can be more. Our technology is helping surgeons make smarter decisions with a new approach to better address knee pain and extend active lifestyles, ultimately raising the standard of care for today’s changing patient population.”
The OvertureTi Knee Resurfacing System is available for immediate use. All instrumentation is sterile-packed for streamlined, single-use efficiency in the operating room, the company said.
Overture Orthopaedics®, OvertureTi Knee Resurfacing System®, and Focalplasty® are trademarks of Overture Orthopaedics.
Hydrophilic Coating Can Be Customized to Optimize Performance for Specific Device Requirements
Freudenberg Medical is working to redefine hydrophilic coatings by uniting advanced chemistry, device engineering, and customer-first partnerships.
BEVERLY, Mass.—As a provider of hydrophilic coatings, Freudenberg Medical prides itself on uniting advanced surface chemistry with deep, in-house medical device design and manufacturing capabilities. In doing so, the company brings together complementary technologies “to enhance manufacturability and deliver greater productivity and reliability across the entire product development lifecycle,” according to a release from Freudenberg Medical.
The company highlighted these abilities recently with its launch of LUBRITEQ™, a new high-performance hydrophilic coating, along with a comprehensive suite of associated development and manufacturing services. LUBRITEQ is now available globally across Freudenberg Medical’s vertically integrated operations,” the release stated.
“Hydrophilic coatings and device design must work in perfect harmony to deliver optimal performance, yet the industry has traditionally approached them as separate disciplines,” said Michael McGee, CEO of Freudenberg Medical, in a statement. “As a uniquely integrated technical partner, we enable customers to move faster, with less risk and greater confidence, by combining product design, advanced materials, precision coatings, and application expertise for seamless development, scale-up, and launch.”
Deep expertise in surface chemistry
Freudenberg Medical is a global contract design and manufacturing (CDMO) partner to the medical device and biopharma industry. The company’s more than 25 years of experience in coating science is supported by what it called “a proven legacy of innovation, including expertise integrated through Hemoteq, that has enabled customers to achieve multiple industry firsts in complex drug-device and surface modification solutions.”
“With more than 40 coating scientists, chemical engineers, and technicians working alongside 250 medical device and catheter engineers globally, we have unparalleled expertise in developing complex chemistry and drug-device solutions for our customers,” said McGee. “We already support over one million medical devices each year for functional surface coatings, making the introduction of LUBRITEQ™, our hydrophilic coating solution, a natural evolution and logical next step aligned with our customers’ needs.”
Shaped by customer needs
Freudenberg Medical’s decision to enter the hydrophilic coatings market was guided by extensive engagement with customers and industry stakeholders to understand where existing solutions and partnerships can be improved, and where unmet needs continue to exist. Through these exchanges, it became clear that while coating technologies have advanced, customer needs are not being fully served—particularly when it comes to the flexibility of support services and complexities of royalties, licenses, and fees, the company said.
“We heard consistent feedback that directly shaped our strategy,” said Freudenberg Medical Chief Commercial Officer Keith Kiernan, in the release. “Customers aren’t just looking for a better coating—they’re looking for a better partner to de-risk their product launches. We deliberately set out to deliver a solution—and a partnership model—that reflects exactly what customers told us they need. No royalties, no rigidity—just coating performance, partnership, and progress.”
LUBRITEQ is described as “an advanced, UV-curable, single-step hydrophilic coating engineered for versatility and performance across a wide range of medical device applications and polymer and metal substrates.” The coating is said to provide high lubricity with exceptional durability, consistent uniformity, and ultra-low particulates. It is customizable to optimize performance for specific device requirements and is reportedly compatible with “virtually all approved materials used in medical device designs,” the release said.
Recognizing that coating performance alone is not enough, Freudenberg Medical is launching LUBRITEQ alongside a comprehensive services model that is said to support customers across the full product lifecycle—from early feasibility through commercial production. These services are reported to include a rapid two-week first feasibility turnaround, tailored coating customization programs, and flexible volume-manufacturing options, including process transfer to the customer or contract coating support across Freudenberg Medical’s global site network.
“This integrated approach eliminates hand-offs across multiple suppliers, reducing risk and accelerating product development timelines,” the release stated.
Customer-first and transparent commercial model
The company said that in contrast to traditional industry practices, it is introducing a straightforward and transparent commercial model for LUBRITEQ that is designed to eliminate hidden costs and reduce program launch risk. “Customers can access a high-performance hydrophilic coating solution with no royalties, no fixed or annual fees, and no regulatory file access fees,” according to the release.
Together, these elements are said to remove commercial barriers and enable long-term, trust-based partnerships.
“With LUBRITEQ™, Freudenberg Medical is raising the bar for what a hydrophilic coating partner can and should be,” said CEO Michael McGee, in the release. “The early responses we’re seeing from customers confirm that we’re meeting a real need. Great technology, great service, and a commercial model built on trust—that’s the standard we are setting.”
Freudenberg Medical offers an array of vertically integrated capabilities that are reported to include minimally invasive solutions, such as complex catheters, hypotubes, steerable shafts, and handles. The company’s capabilities also include drug and hydrophilic coatings, precision molding, two-shot and micro molding, advanced extrusions, and solutions for smart medical devices, the release said.
Next-Generation GPS Shoulder System Expands Advita Ortho’s Surgical Navigation Platform
GAINESVILLE, Fla.—A next-generation GPS Shoulder system developed by medical technology company Advita Ortho is reported to expand the company’s navigation platform with an updated station and software designed to deliver a faster, more intuitive user experience for surgical teams.
In a January release, Advita Ortho stated that the new GPS Shoulder system offers a sleek interface and high-resolution touchscreen. It is designed to support implant placement accuracy, reduce complications, and improve clinical outcomes, the company said.
Surgeon Howard Routman, D.O., performed the first surgical cases using the new system at JFK Medical Center in Atlantis, Florida.

Advita Ortho has expanded its navigation platform with the next-generation GPS Shoulder system. It features an updated station and software designed for a faster, more intuitive surgical experience, the company said. (Image courtesy Advita Ortho, PRNewswire)
“These cases were an opportunity to see the latest GPS technology in action, and how its enhancements can raise the bar for efficiency and intraoperative 3D feedback in real time,” Routman said in a statement. “The impact was clear, even before we began to navigate the case. The upgrades allowed us to better meet our goals of enhancing accuracy, reducing variability, and delivering optimal results with faster processing speed, resulting in a reduction in surgical time. The GPS platform is moving even closer to becoming the standard of care.”
Today, more than half of Advita’s global shoulder cases are said to be performed using GPS, reflecting surgeons’ growing confidence in the company’s advanced navigation system.
The system offers a variety of key features, beginning with reusable active trackers that integrate into hospital and ASC workflows using existing instrumentation sterilization processes. This helps reduce per-case costs versus disposable tracker systems, the company said in the release.
In addition, calibration checks are performed in seconds, capturing real-time data without disrupting surgical flow. System features also include wide angular visibility with a 135-degree field of view and real-time auxiliary visual connectivity, and postoperative reporting tools that organize case data for review, documentation and analysis.
“With this release, we’ve taken everything surgeons love about GPS and elevated the experience even further,” said Advita Ortho CEO Aurelio Sahagun, in a statement. “This system reflects the trust surgeons place in our GPS technology. By delivering a more seamless and enhanced experience, we’re helping surgeons focus on what matters most: patient care.”
As part of the company’s Active Intelligence® ecosystem, GPS connects preoperative planning tools, real-time intraoperative analytics, and postoperative insights to support surgeons across the entire episode of care.
According to Advita Ortho, the next-generation GPS Shoulder system is being introduced through limited release. “The GPS technology is available to Advita Ortho customers at no capital cost,” the release stated.
Advita Ortho describes itself as “a global medical device company committed to advancing patients’ lives with high-quality implants and surgical technologies.”
“We specialize in high-quality implants and a comprehensive suite of integrated surgical technologies that are powered by data, artificial intelligence, and machine learning,” the company said in the release.
Surgeon Completes First GPS-Guided Shoulder Case with Central Screw Baseplate
According to Advita Ortho, the milestone marks the first clinical use of navigation to support central screw placement in shoulder arthroplasty.
GAINESVILLE, Fla.—Global medical technology company Advita Ortho recently announced the successful completion of what it described as “the first surgical case using Advita GPS™ in combination with the Central Screw Baseplate (CSB).”

Advita GPS™ supports Central Screw Baseplate placement during the first GPS-guided shoulder arthroplasty case, representing the only technology designed to provide guidance for this procedure. (Image courtesy Advita Ortho, PRNewswire)
The procedure, performed by fellowship-trained shoulder surgeon Andrew S. Neviaser, M.D., is reported to represent the first clinical use of navigation technology to assist with central screw placement in shoulder arthroplasty.
“The CSB is one of the most versatile glenoid solutions designed for use in both primary and complex revision cases,” according to a release from Advita Ortho. “Engineered to support fixation in neutral and augmented configurations, the CSB implant is often used in challenging shoulder procedures. To further support precision in central screw placement, GPS Shoulder, the only navigation platform designed specifically for this application, provides surgeons with real-time feedback on implant positioning and screw trajectory.”
When using the Central Screw Baseplate in combination with Advita GPS, surgeons can “leverage 3D preoperative planning with intraoperative navigation to visualize patient anatomy through real-time procedural guidance,” the release stated.
“Revision shoulder arthroplasty presents unique challenges, especially when bone quality or anatomy has been compromised,” Neviaser said in a statement. “GPS Shoulder provides real-time intraoperative guidance for central screw trajectory and baseplate positioning, while helping us better match the implant to each patient’s unique anatomy for a more precise fit.”
The GPS Shoulder system and Central Screw Baseplate are designed to complement the Equinoxe® Reverse Shoulder System, which is reported to support a broad range of anatomic and clinical needs with modular fixation options. Together, 3D planning and navigation are said to support visualization and decision-making in cases where complex anatomy or revision scenarios may challenge traditional techniques.
“This first case reflects our commitment to solving real clinical challenges through meaningful innovation,” said Laurent Angibaud, senior vice president of advanced surgical technologies at Advita Ortho, in a statement. “By continuing to integrate our implant portfolio with enabling technologies, GPS Shoulder is gaining momentum among Advita shoulder surgeons and increasingly establishing itself as a standard-setting navigation solution.”
According to Advita Ortho, the announcement reinforces the company’s focus on developing data-driven technologies that are designed to support surgeons across the full spectrum of total joint replacement procedures.
AI-enabled Technology Reported to Help Surgeons Focus on Clinical Factors that Matter Most in Joint Replacement Procedures
Advita Ortho’s patented surgical planning technology uses a weight-based algorithm to prioritize different factors based on patient anatomy and surgeon preference.
GAINESVILLE, Fla.—Surgical planning for joint replacement planning requires surgeons to assess numerous factors, such as implant alignment, sizing, and soft-tissue balance, and often requires trade-offs to achieve the best possible outcomes. But as personalized approaches become more prevalent, the number of variables involved in surgical planning continues to increase, making decision-making more complex.
Advita Ortho is a global medical technology company working in this environment. The company was recently granted a U.S. patent (#12,544,141) by the U.S. Patent and Trademark Office for an AI-enabled surgical planning framework that is “designed to help surgeons focus on the clinical factors that matter most during joint replacement procedures,” according to a company release.
The patent is said to mark a significant milestone in Advita’s long-term strategy to advance smarter, more personalized orthopedic care.
“This patent represents an important step in our journey toward smarter surgical planning,” said Laurent Angibaud, senior vice president of advanced surgical technologies at Advita Ortho, in a statement. “Surgeons already make nuanced decisions based on experience and philosophy. Our goal is to support that thinking with intelligent tools that adapt to the surgeon, the patient, and the procedure, while preserving clinical control.”
According to Avita Ortho, its patented approach outlines a weight-based algorithm to help prioritize these variables, rather than treating all parameters equally. By establishing a framework for how different factors may be balanced based on patient anatomy and the surgeon preference, the patent defines a pathway toward more consistent planning decisions aligned with individual surgical philosophies. Over time, this framework is designed to learn from previous cases and refine its recommendations as more data becomes available.
“This work reflects how we are applying artificial intelligence in practical, surgeon-driven ways,” said Aurelio Sahagun, CEO at Advita Ortho, in the release. “By combining clinical insight with real-world data, we are building AI technologies that help improve consistency, personalization, and confidence in surgical decision-making.”
By defining how priorities are handled during surgical planning, the patent is said to establish a foundation for future innovation across Advita’s Active Intelligence® ecosystem, including decision support, data-driven optimization, and visualization tools.
